Biogen's Aduhelm rejected by EMA committee
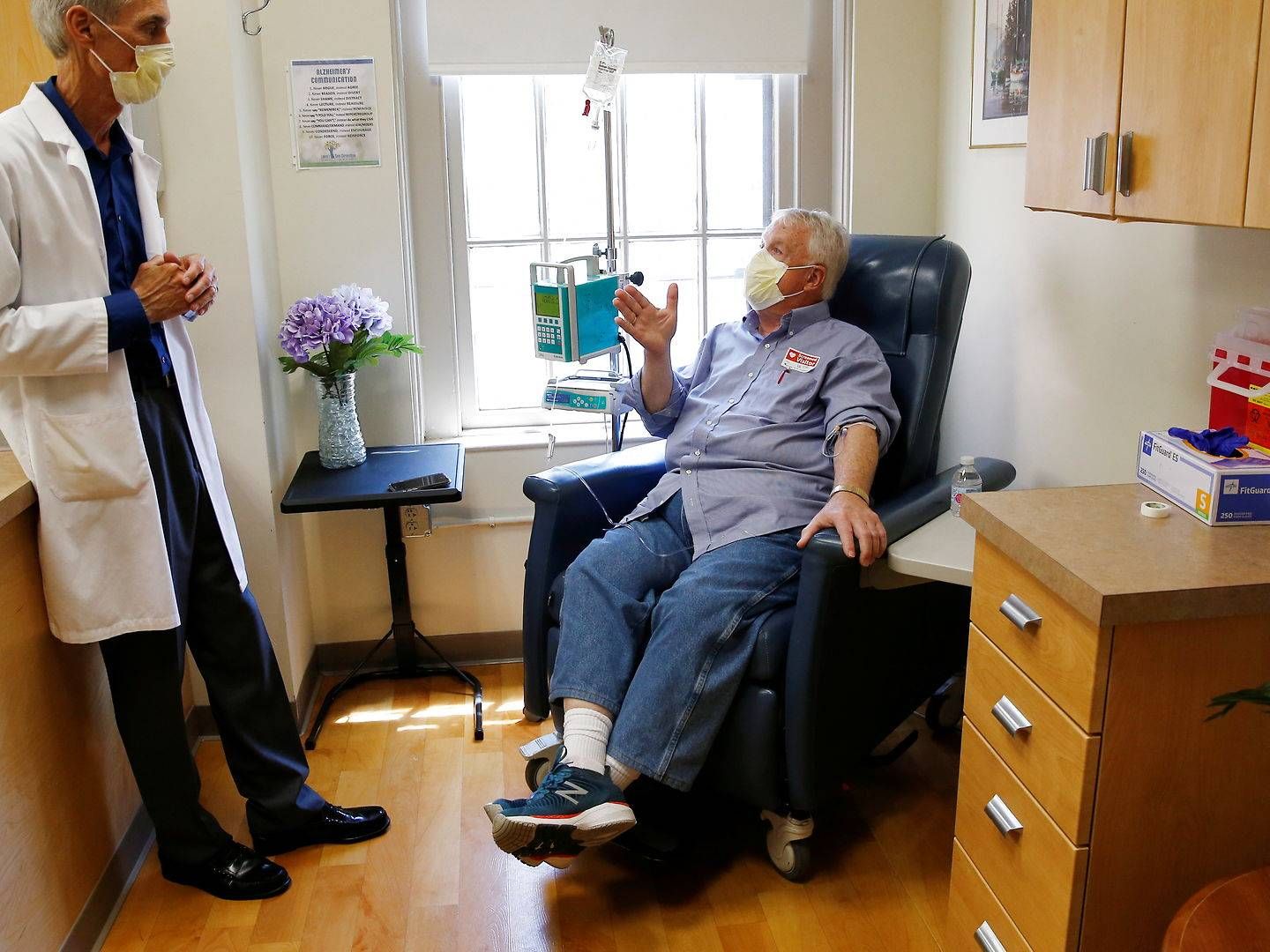
On Friday, Alzheimer's drug Aduhelm, which pharmaceutical firm Biogen is behind, has been rejected by the European Medicines Agency's (EMA) Committee for Medicinal Products for Human Use (CHMP).
Read the whole article
Get access for 14 days for free. No credit card is needed, and you will not be automatically signed up for a paid subscription after the free trial.