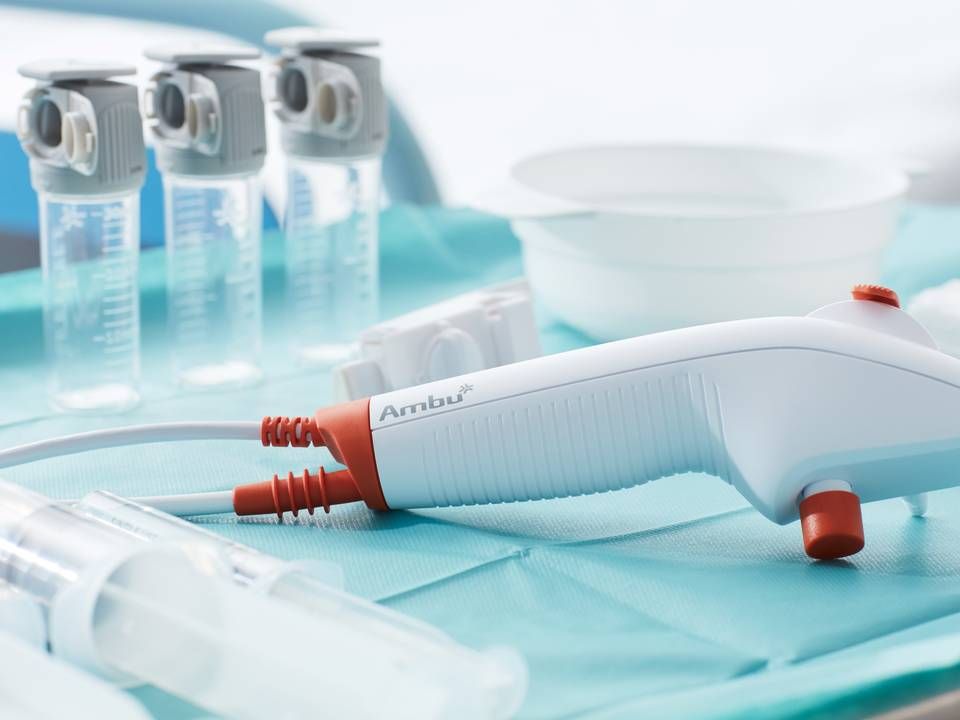
Ambu gets EU approval for bronchoscope
The EU has approved Ambu's latest bronchoscope and accompanying sample collector, the Ambu Ascope 4 Broncho Sampler Set. It has received the CE-mark authorization from the authority.
Read the whole article
Get access for 14 days for free. No credit card is needed, and you will not be automatically signed up for a paid subscription after the free trial.
With your free trial you get:
Get full access for you and your coworkers
Start a free company trial todayRelated articles
Pentax rejects Ambu's claims of head start
For subscribers
Ambu launches new duodenoscope in four weeks
For subscribers
Ambu joins green climate initiative
For subscribers